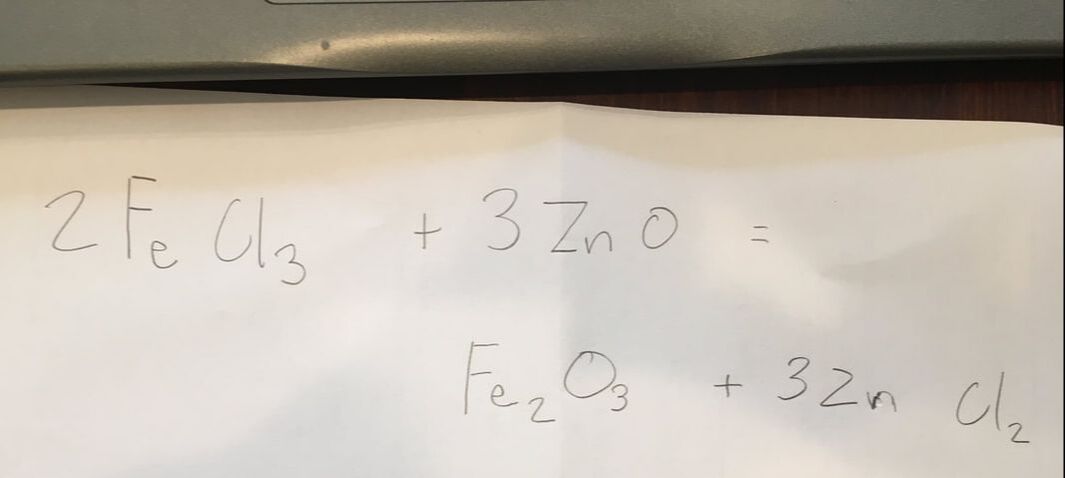The Task:
Vinny, Chris, Cooper, and I were tasked with creating an art piece using chemistry. We had to create four pieces and explain the chemistry behind each one, in our case the equations and chemistry behind each color of paint we created. Each piece shared a general theme, in our case the ocean. Certain artwork will be selected to be shown at the opening of the new STEM building. In this project, my group worked as a unit but did most of the tasks individually. Not only did we each make a different color but we also made four different pieces of art. We made a series of themed paintings and presented the art to our class.
Vinny, Chris, Cooper, and I were tasked with creating an art piece using chemistry. We had to create four pieces and explain the chemistry behind each one, in our case the equations and chemistry behind each color of paint we created. Each piece shared a general theme, in our case the ocean. Certain artwork will be selected to be shown at the opening of the new STEM building. In this project, my group worked as a unit but did most of the tasks individually. Not only did we each make a different color but we also made four different pieces of art. We made a series of themed paintings and presented the art to our class.
The Messy Business of Chemistry:
I decided to make "Red Ochre" a reddish brown color. The Chemicle formula of "Red Ochre" is "Fe2O3"
In order to create this I combined Zinc Oxide and Iron(III) Chloride.
In order to create this I combined Zinc Oxide and Iron(III) Chloride.
After Mixing these two chemicals and waiting for the double displacement reaction I then filtered out the precipitate.
As you can see this beaker says CaO because at first I used calcium oxide instead of zinc oxide but the reaction was not as good.
I then put this precipitate in paint base and made my "Red Ochre".
Important Concepts:
Ion - An ion is an atom that doesn't have a neutral charge. It can be either a cation with a positive charge or an anion with a negative charge.
Decomposition Reaction - In a decomposition reaction one, multi atomic, reactant seperates: AB > A + B
Synthesis Reaction - In a synthesis reaction, two reactants create one product: A + B > AB
Single Displacement Reaction - In a single replacement reaction, a reactant switches out either its anion or cation: A + BC > AC + B
Combustion Reaction - In a combustion a hydrocarbon reacts with oxygen and heat to create water and carbon dioxide.
Double Displacement Reaction - In a double replacement reaction the two reactants switch cations: AB + CD > AD + BC
Precipitate - A precipitate is a solid formed from a double displacement reaction
Red Ochre - A pigment formed from the precipitate of Iron Oxide
Ion - An ion is an atom that doesn't have a neutral charge. It can be either a cation with a positive charge or an anion with a negative charge.
Decomposition Reaction - In a decomposition reaction one, multi atomic, reactant seperates: AB > A + B
Synthesis Reaction - In a synthesis reaction, two reactants create one product: A + B > AB
Single Displacement Reaction - In a single replacement reaction, a reactant switches out either its anion or cation: A + BC > AC + B
Combustion Reaction - In a combustion a hydrocarbon reacts with oxygen and heat to create water and carbon dioxide.
Double Displacement Reaction - In a double replacement reaction the two reactants switch cations: AB + CD > AD + BC
Precipitate - A precipitate is a solid formed from a double displacement reaction
Red Ochre - A pigment formed from the precipitate of Iron Oxide
Reflection:
This project was an eye opener for how many different forms art can take. I think I did a good job being open to peoples ideas and also persisting through challenges with our project. Half way through our project I discovered Calcium Oxide did not work and that I had to change to Zinc Oxide. I could, however, still improve. I found myself dealing with some hard collaboration issues as well as struggling with the changing variables of our project these are things that are vital to deal with in order to have success with any project and it is something I will strive to work on.